|
Some miscellaneous facts on the first law of thermodynamics However, heat can be transferred from one place to another, and this sort of energy is only convertible from one form to another. 
Well, thermodynamic processes are there that always consider a state i.e., conservation of energy. This law states that heat is one type of energy that cannot be produced or neither be ruined. The third law of thermodynamics in terms of entropy 
The second law of thermodynamics in terms of entropy The first law of thermodynamics relating to entropy We will now understand the three laws associated with thermodynamics. In this article, we will know about two definitions of entropy, such as thermodynamic definition as well as statistical definition. Many definitions are associated with entropy. Entropy is a measurement of disorder or randomness found in a system. In 1850, Rudolf, a German physicist, named its entropy. The applications of entropy are useful in thermodynamic stages. Entropy can be explained in numerous ways. 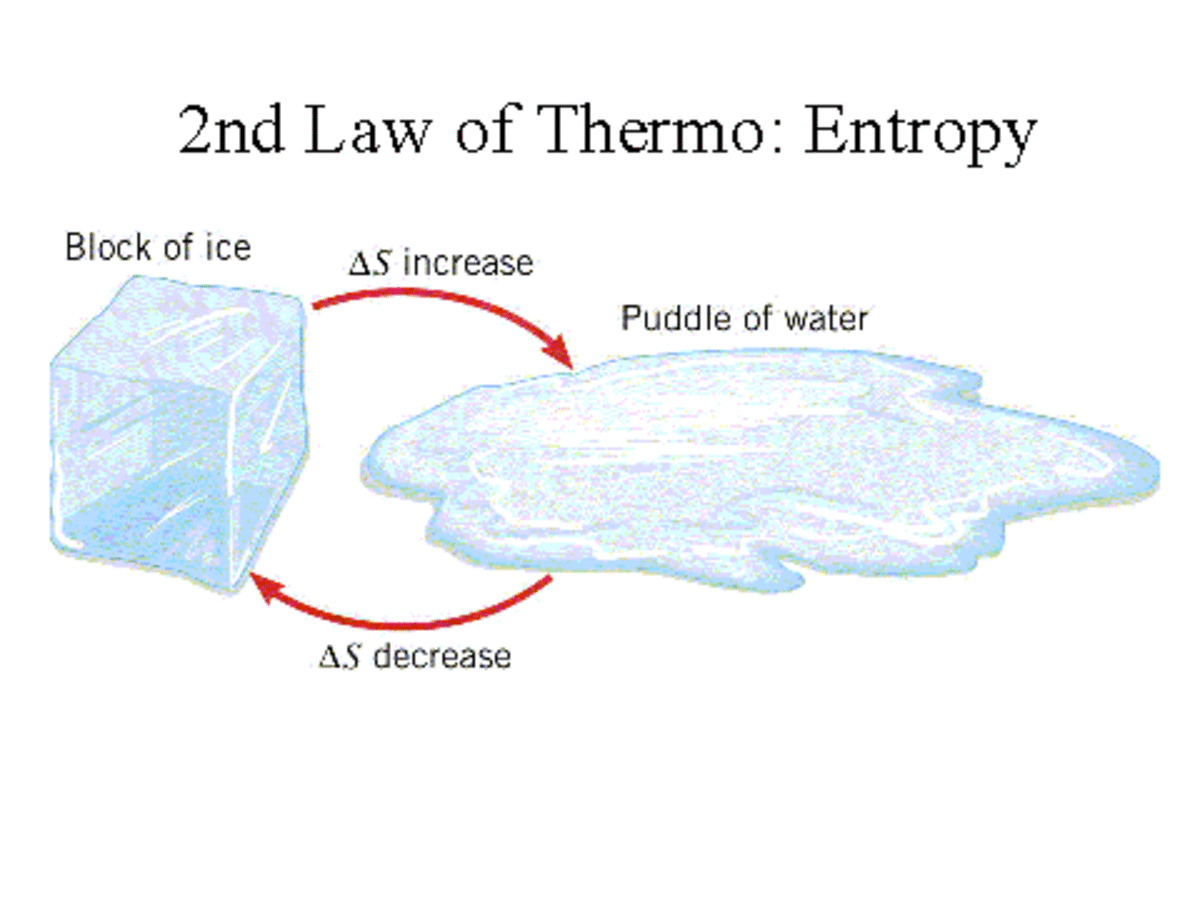
Students should understand the importance of entropy law as it is very helpful for studying physics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
